Pfizer Applies For Emergency Federal Approval Of Its COVID-19 Vaccine
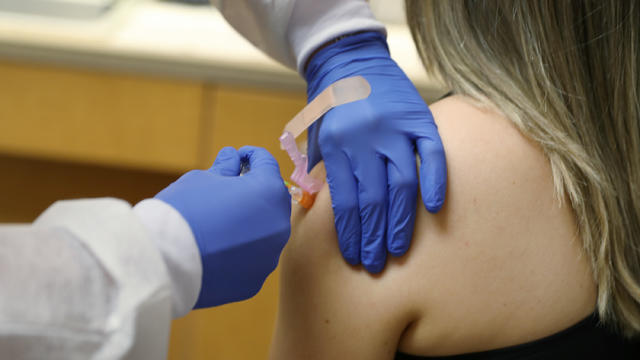
Pfizer argues protection plus a good safety record means the vaccine should qualify for emergency use authorization, something the FDA can grant before the final testing is fully complete.