
Michigan man found dead after driving off ramp into St. Clair River
A Michigan man was found dead after driving his car off a ramp and into the St. Clair River in Clay Township, according to the St. Clair County Sheriff's Office.
Watch CBS News

A Michigan man was found dead after driving his car off a ramp and into the St. Clair River in Clay Township, according to the St. Clair County Sheriff's Office.

The Oakland County Sheriff's Office is investigating after a 22-year-old woman was found shot to death on a sidewalk in Pontiac on Sunday.

Reform-minded candidates won several races as members of the United Auto Workers union voted on their leaders in an election that stemmed from a federal bribery and embezzlement scandal involving former union officials.

More than 35 classic vehicles are being donated to Northwood University, worth more than $2 million.

Presidential candidates in 2024 will be putting more attention onto Michigan following a vote by a DNC panel.

Detroit police are looking for a man who they say lit a garbage dumpster on fire and tried to spread that fire to a nearby store.

After a week of silence from former Marine and Novi man Paul Whelan, his brother sets his sight on getting him home.

The Detroit Lions (4-7) hope to rebound from a Thanksgiving Day loss as the team prepares to host the Jacksonville Jaguars (4-7).

The Red Hook coffee shop is opening its fourth location in Detroit's Midtown neighborhood.

Michigan State University announced that its teacher preparation program will shorten, moving from a five-year program, to a four-year one.

Crews will be closing lanes on 8 Mile Road near Telegraph as they work to relocate underground and overhead utilities, according to the Michigan Department of Transportation.

Prosecutors say Warren police were notified last month of Greenday Massage allegedly conducting sexual favors and prostitution. Two undercover officers made appointments at the parlor and were allegedly offered sexual favors for money.

A Michigan woman is competent to stand trial for her role in the death of her 15-year-old son, who was allegedly subjected to ice baths while malnourished, a judge has ruled.

Anyone interested in reserving a laptop or hotspot can do so at any Detroit Public Library location or over the phone with a library card. It is free to reserve one of these items, but they must be reserved in advance and must be returned after 90 days.

Police are investigating a student at Ecorse High School after they brought a gun to school, according to the Ecorse Public Schools Superintendent Josha Talison.
Fire officials are investigating after a man was injured in an Ann Arbor house explosion Monday morning. CBS News Detroit's Jordan Burrows gives the latest updates from the scene.

The Michigan State Spartans celebrated a weekend of commencement ceremonies.

Families poured in to check out this Michigan treasure.

The City of Detroit's Easter Fun Fest returns to provide families an opportunity to not only get outside and be active, but ring in the holiday with some fun.

Mixed media artist Donald Calloway has been creating art in some form or fashion most of his life. His art studio takes you on a journey, as his creative collection of his art runs the gamut.

Soon after a positive test, Dorfman found himself hospitalized, on a ventilator and in a medically induced coma.
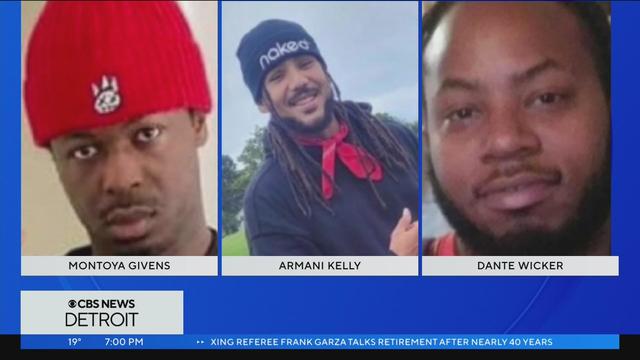
It has been 12 days since anyone has seen Armani Kelly, Montoya Givens and Dante Wicker. Now, a Facebook live video conversation has surfaced where Kelly is seen talking with three men about coming to Detroit only two days before he went missing.

A Plymouth man's cancer diagnosis hit the reset button on how he lived his life. Since then, he and his family have been giving back to others facing the same struggles.

The Sphinx organization gives a platform to Black and Latinx performers of all ages.