FDA Approves QWO Enzyme Injection That Dissolves Cellulite
NEW YORK (CBSNewYork) -- Cellulite is very common and extremely challenging to get rid of.
But the Food and Drug Administration has just approved an injection that actually dissolves the cellulite, CBS2's Dr. Max Gomez reported Wednesday.
Over the years there have been a number of treatments that purport to get rid of cellulite. There have been creams and ointments, radio-frequency devices, lasers that actually work and even suction gadgets.
Now, there's an injectable enzyme that the FDA says actually does the job.
FLASHBACK: Dermatologists Turn To Cellulite Treatment To Clear Up Acne Scars
Like almost 80-90% of women, Tina Marie Williams had cellulite and she did not like it.
"It was in the butt area, in the back of the thigh. It may have kind of a dent type of a look to it. I wanted better," said Tina Marie Williams, a cellulite patient.
So when Williams heard of a clinical trial at JUVA Skin & Laser Center that was almost non-invasive for cellulite, she signed up.
"QWO, basically, it's an enzyme and the enzyme dissolves the fibrous brands," said Dr. Bruce Katz of JUVA Skin & Laser Center.
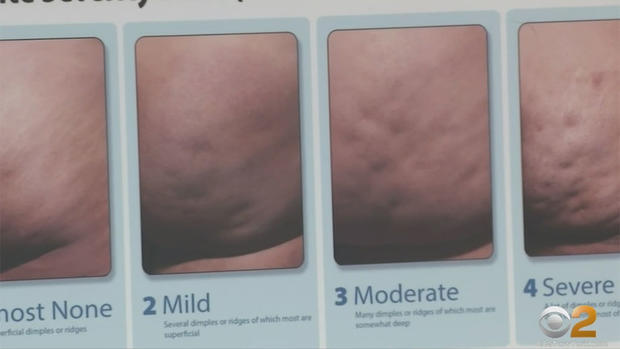
Dr. Katz led one of the clinical trails into the enzyme he calls QWO, pronounced "Kwo." Cellulite is caused when tiny fibrous bands that connect the under side of the skin to a deeper layer contract, pulling the skin down into those unsightly dimples that can vary in severity, but almost always occur on the buttocks and thighs.
Three small injections of a type of collagenase enzyme are aimed right at the tiny band under each dimple to dissolve it and the skin lifts back up. The results are quite noticeable, but will require multiple treatments.
"Once every three weeks, a total of three times, and we're actually doing a five year follow-up just to see that this treatment does endure," Dr. Katz said.
"It was, absolutely. It was worth it," Williams said.
The main side effects were a little bruising and soreness at the injection sites. As for cost, the company has not yet announced pricing, but that same enzyme used for other conditions has been very expensive. The FDA approval right now is for the buttocks. Approval for thighs is pending.
If you are interested in this new injection, the company said you'll have to wait until next spring as it ramps up production.