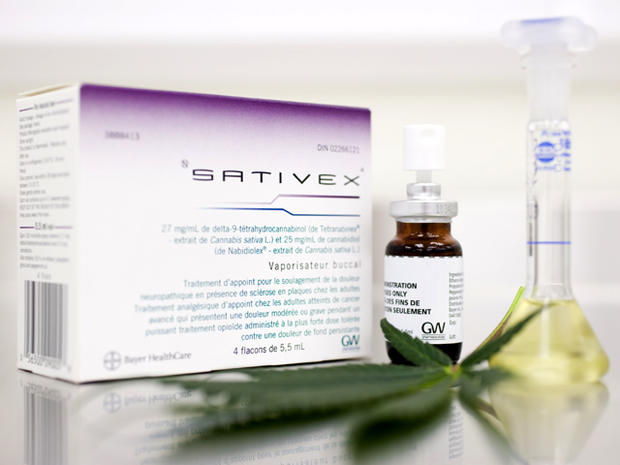
Marijuana mouth spray seeks FDA approval as painkiller
(CBS/AP) A marijuana-based mouth spray may get FDA approval as soon as 2013 - at least that's what British manufactuer GW Pharma hopes. The company is in advanced clinical trials on the world's first pharmaceutical developed from raw marijuana plants.
Other marijuana-based drugs currently on the market use synthetic equivalents of pot, but this stuff's made from the real deal. Its makers want to market the drug in the U.S. as a treatment for cancer pain.
The spray, called Sativex, contains marijuana's two best known components - delta 9-THC and cannabidiol. The medication has already been approved in Canada, New Zealand and eight European countries for relieving muscle spasms associated with multiple sclerosis.
If the FDA approves the spray, it would represent an important milestone in the U.S., which has had a sticky history with medical uses of marijuana. Currently, 16 states and Washington D.C. allow residents to legally use medical marijuana with doctors' recommendations. The U.S. Drug Enforcement Administration considers pot a dangerous drug with no medical value, subjected it to federal prosecution. But the availability of a chemically similar prescription could increase pressure on the federal government to revisit its position.
"There is a real disconnect between what the public seems to be demanding and what the states have pushed for and what the market is providing," said Aron Lichtman, a Virginia Commonwealth University pharmacology professor and president of the International Cannabinoid Research Society. "It seems to me a company with a great deal of vision would say, 'If there is this demand and need, we could develop a drug that will help people and we will make a lot of money.'"
Possessing pot is also still illegal across the pond in the U.K., but about a decade ago GW Pharma's founder, Dr. Geoffrey Guy, received permission to grow it to develop a prescription drug. Guy proposed the idea at a scientific conference that heard anecdotal evidence that pot provides relief to multiple sclerosis patients, and the British government welcomed it as a potential way "to draw a clear line between recreational and medicinal use," company spokesman Mark Rogerson said.
"We were the first ones to charge forward and a lot of people were watching to see what happened to us," Rogerson said. "I think we are clearly past that stage."
In 1985, the FDA approved two pills containing synthetic THC, Marinol and Cesamet, to ease side-effects of chemotherapy in cancer patients. The agency eventually allowed Marinol to be prescribed to stimulate the appetites of AIDS patients. The drug's patent expired last year, and other U.S. companies have been developing formulations that could be administered through dissolving pills, creams and skin patches and perhaps be used for other ailments.
Doctors and people with multiple sclerosis patients are cautiously optimistic about Sativex. The National Multiple Sclerosis Society has not endorsed marijuana use by patients, but the organization is sponsoring a study by a University of California, Davis neurologist to determine how smoking pot compares to Marinol in alleviating painful muscle spasms.
Interest in new and better marijuana-based medicines has been building since the discovery in the late 1980s and 1990s that mammals have receptors in their central nervous systems, several organs and immune systems for the chemicals in botanical cannabis and that their bodies also produce natural cannabinoids that work on the same receptors.
One of the first drugs to build on those breakthroughs was an anti-obesity medication developed by Sanofi Aventis called Acomplia that blocked the same chemical receptors that trigger the munchies in pot smokers. Acomplia was approved throughout Europe and heralded as a possible new treatment for smoking cessation and metabolic disorders that can lead to heart attacks.
The FDA was reviewing its safety as a diet drug when follow-up studies showed that people taking the drug were at heightened risk of suicide and other psychiatric disorders. Sanofi pulled it from the market in late 2008.
Given that drug companies already were reluctant "to touch anything that is THC-like with a 10-foot- pole," the setback had a chilling effect on cannabinoid drug development, according to Lichtman.
He said, "Big companies like Merck and Pfizer were developing their own versions (of Acomplia), so all of those programs they spent millions and millions on just went away..."