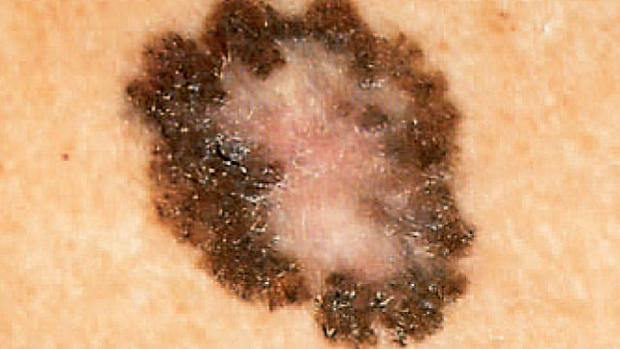
FDA approval of melanoma drug called "huge step" in cancer care
(CBS/AP) The FDA has approved a first-of-a-kind drug for melanoma, the deadliest form of skin cancer. Zelboraf works by targeting a mutant gene that's found in about half of melanoma patients.
PICTURES - Is it skin cancer? 38 photos that could save your life
"Zelboraf is a huge step forward in the fight against metastatic melanoma, and the advancement of treatment for the deadliest form of skin cancer," Dr. Anna Pavlick, director of the melanoma program at the New York University Cancer Institute and one of the scientists involved in research on the drug.
Melanoma is the fastest-growing form of cancer in terms of new diagnoses. Scientists attribute the acceleration to longer life expectancies in the U.S. and increased indoor tanning by the young.
About 68,000 people in the U.S. were diagnosed with melanoma last year and 8,700 died, according to the American Cancer Society.
Melanoma has been considered one of the toughest cancers to treat, with few drug options. In March the FDA approved a Bristol-Myers Squibb drug, Yervoy, which was the first drug shown to prolong survival in patients with advanced skin cancer.
Zelboraf will provide a second option for melanoma patients with a mutated form of a protein called BRAF that helps with cell growth when working normally. Zelboraf slows tumor growth by blocking the mutated form of the protein.
The FDA approved the drug on the basis of a 675-patient study in which patients received either Zelboraf or a chemotherapy drug. Among people in the ongoing study, 77 percent on Zelboraf are alive versus 64 percent of those taking the older drug, the FDA said.
Despite the higher survival rate, melanoma adapts quickly, and patients saw their tumors resume growth after seven months, on average.
Side effects with the drug included skin rashes, joint pain, fatigue, diarrhea and hair loss. About 26 percent of patients developed a less serious form of skin cancer.
Melanoma patient advocates praised the FDA for clearing the drug well ahead of an Oct. 28 target date to complete its review.
"The FDA's quick action on this drug approval is important because it gives melanoma patients a new way to fight this deadly disease," Timothy Turnham, director of the Melanoma Research Foundation, said in a statement.
Zelboraf will be available within two weeks, according to its maker, Roche.
The National Cancer Insitute has more on melanoma.