Ozone layer hits a "significant milestone" as concentrations of harmful chemicals drop 50%, NOAA says
Earth's ozone layer is vital to protecting all forms of life – from crops to humans – from the sun's harmful radiation. This shield in Earth's stratosphere has been depleted for decades, putting life on the planet at risk, but new research from NOAA says that it may now have a chance to at least partially recover.
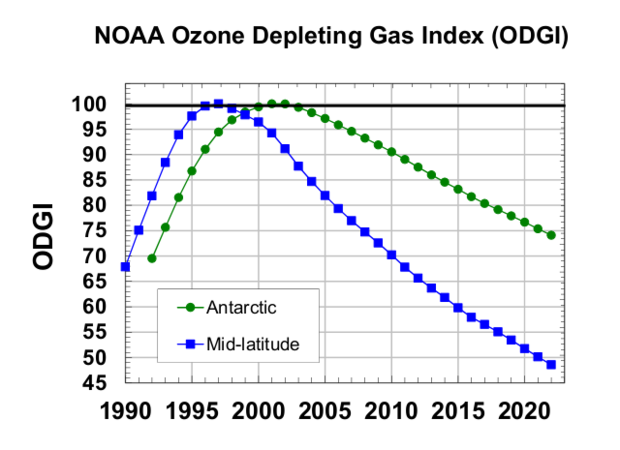
In new research, NOAA found that global concentrations of the harmful chemicals that damage the ozone layer have declined just over 50% in the mid-latitude stratosphere, to levels observed in 1980. The continued decline, NOAA scientists said, "shows the threat to the ozone layer receding below a significant milestone in 2022."
Though slower, there has also been a decline in concentrations over Antarctica, where a hole in the ozone layer appears every year. NOAA found concentrations have declined 26% since peak values in the region in the '90s. In 2021, that hole was larger than usual – bigger than the size of Antarctica itself, but now NOAA says that the Antarctic ozone layer is projected to recover "sometime around 2070."
International regulations and compliance for managing those chemicals are the reason for the "slow but steady" three-decade progress, the agency said.
Stephen Montzka, senior scientist for NOAA's Global Monitoring Laboratory, said the progress is great, but "at the same time, it's a bit humbling to realize that science is still a long way from being able to claim that the issue of ozone depletion is behind us."
Scientists have been closely monitoring the ozone since the '80s, when it was discovered that certain man-made chemicals were "seriously damaging" Earth's vital protective layer. In 1987, just seven years after ozone depletion from chemicals was made most obvious, every country on Earth – for the first and only time ever – ratified a treaty, known as the Montreal Protocol, to regulate the chemicals to protect Earth.
Among those man-made chemical compounds are chlorofluorocarbons (CFCs), which people started using in the '60s in air conditioners, aerosol spray cans, styrofoam and industrial cleaning products, according to the University Corporation for Atmospheric Research.
Once used, the CFCs make their way up to the stratosphere, where ultraviolet radiation breaks up the compounds and releases chlorine atoms, which along with bromine, is dangerous to the ozone, according to the EPA. These atoms are known for destroying ozone molecules – just one chlorine atom can destroy more than 100,000 ozone molecules, the agency said, adding that with these atoms, the ozone "can be destroyed more quickly than it is naturally created."
Hydrochlorofluorocarbons (HCFCs) were developed as a temporary alternative to CFCs, as they have a shorter atmospheric lifetime compared to CFCs and don't give as much reactive chlorine to the stratosphere. However, they still have the capability to "destroy stratospheric ozone," according to NOAA, and production in developed countries was prohibited in 2020.
And while the success so far is promising, scientists said, the fight isn't over.
"Ozone layer recovery is not a forgone conclusion," they said in their report. "Full recovery is expected only with sustained declines in atmospheric chlorine and bromine in future years and continued adherence to the production and consumption restrictions outlined in the Protocol."