Fake cancer drug surfaces again from overseas
(CBS News) It is just about the worst kind of fraud you can imagine -- fake cancer drugs, not much more than a vial of water with a drug label on it. CBS News previously reported on this, but it's happened again: A number of clinics received counterfeit vials of cancer drugs. CBS News chief investigative correspondent Armen Keteyian has traced the source overseas.
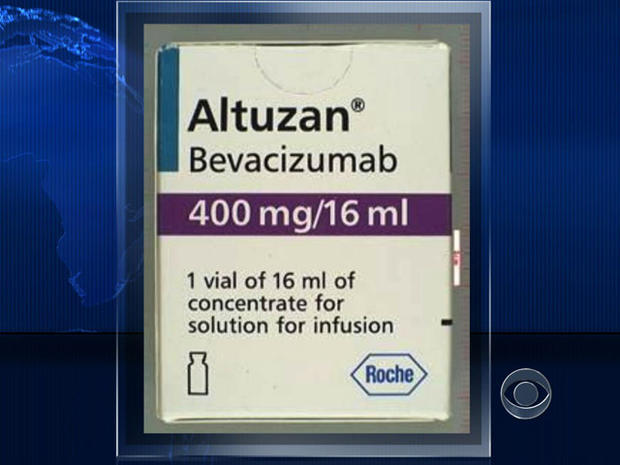
This week, the Food and Drug Administration alerted healthcare professionals that 120 vials of fake Altuzan -- which contained no active ingredient -- had entered the U.S from distributors in the U.K. after being purchased from wholesalers in Turkey.
Connie Jung is with the FDA's Office of Drug Security, Integrity and Recalls. "What we're seeing is a pattern of this risky practice of purchasing unapproved drugs from foreign suppliers," she said.
Altuzan has the same active ingredient as the cancer-fighting Avastin, but is sold only in Turkey and not approved for use in the United States.
According to British authorities, 82 of the counterfeit vials were shipped to the U.S. by River East Supplies, located in the U.K. and owned by Canadian businessman Tom Haughton. Haughton is currently under federal investigation for shipping counterfeit Avastin into the U.S., offering Avastin for about $2,000 a vial -- some $400 less than the manufacturer's price.
We tracked Haughton down last month in Barbados. He denied any wrongdoing and took a strong stance against counterfeits.
Fake Avastin importer claims he broke no laws
Following the trail of fake Avastin
How fake Avastin from overseas ends up in U.S.
"We're depending on the governments and regulators to make sure these supply chains are indeed safe. I will do everything within my power to ensure this never happens ever again."
The FDA said one distributor of the worthless Altuzan was California-based Ban Dune Marketing. In February, its owner pleaded guilty to distributing "adulterated prescription drugs" used for cancer treatment and not approved by the FDA.
According to the government, it offered big discounts on cancer treatment drugs -- "14 to 60 percent" -- requesting doctors keep its price list confidential.
When contacted by CBS News Wednesday, Tom Haughton had no comment. The FDA, meanwhile, would not tell us how many doctors may have received the fake Altuzan, but said these counterfeit products are potentially dangerous to patients who are relying on these medicines to get better.