Asleep At Lab: Whistleblower Allegations From Inside CA's Billion-Dollar COVID Lab
SACRAMENTO (CBS13) — Lab techs sleeping while processing COVID samples for testing. COVID test swabs found in the restrooms. These are just two of the concerning allegations from whistleblowers about what's happening inside the state's new billion-dollar COVID testing lab.
CBS13 uncovered thousands of inconclusive test results coming out of the state's new lab late last year, now whistleblowers want the public to know what's really going on inside.
CBS13 has interviewed more than half a dozen whistleblowers and obtained dozens of internal records and quality control reports.
The documents detail problems ranging from contamination causing inconclusive results to swapped samples and inaccurate results sent to patients.
Records indicate that employees handling patient specimens had not been signed off for competency on crucial skills. Documented competency is required under federal law.
Investigating California's Billion-Dollar Lab
The state's new $25 million lab and $1.7 billion contract with PerkinElmer was supposed to revolutionize COVID testing in California. PerkinElmer is contracted to process up to 150,000 COVID tests daily by March, returning results in 24 to 48 hours.
Data provided by the state indicates that the lab is currently processing an average of fewer than 20,000 tests a day but, according to the state, is being paid the contracted rate for 100,000 tests per day.
In November, CBS13 Investigative Reporter Julie Watts, and her family, were among the first to have their COVID test results processed by the state's new lab. Two out of their four results came back inconclusive.
That's when CBS13 first began asking questions.
ALSO READ: CBS13 Investigation Reveals Inconclusive COVID Results From New State Lab
While the California Department of Public Health would not immediately provide CBS13 with the total number of inconclusive results that were released by the new state lab at the time, Placer County did provide CBS13 with local data. It indicated one out of every 13 Placer County tests processed by the new lab in its first week, came back inconclusive.
In addition to delaying crucial health information amid a pandemic, each inconclusive test, and retest, comes at a cost to taxpayers.
When CBS13 asked California's Health and Human Services Secretary about the inconclusive results during a weekly press briefing, Dr. Mark Ghaly did acknowledge that there had been "a higher number than expected."
Ghaly blamed a chemical reaction.
"One of the validation steps, that confirms the necessary chemical reaction to run the test, didn't occur for some of those inconclusive tests," Ghaly said. "We've identified some of the issues with why they didn't occur and have since corrected it."
CBS13 has now obtained a quality control report from that week confirming Ghaly's explanation.
ALSO READ: Regulators Find 'Significant Deficiencies' At CA COVID Testing Lab – Whistleblowers Respond
Inconclusive COVID Results
However, as CBS13 continued to press the California Department of Public Health (CDPH) for data related to inconclusive tests, the state changed its explanation.
In a January email, the agency pointed to the type of test used at the CDPH-PerkinElmer lab as the reason for the high number of inconclusive results.
They explained that their test is "able to detect lower levels of viral load than other tests on the market." The state added that when low levels are detected "these are reported as inconclusive."
Whatever the reason, in just 3 months, data provided by CDPH indicates that more than 50,000 tests from the state lab did not reveal if the person actually had COVID. That includes samples that CDPH said were reported as inconclusive, invalid, and lost or spilled samples.
Many of those results were released during the peak of the pandemic, between November and the end of the year, as people were traveling and visiting loved ones over the holidays.
Lab directors consulted for this story tell CBS13 that inconclusive results are typically rare.
There are more than 100 COVID labs processing California tests. According to the state, 22 have reported inconclusive results since the start of the pandemic.
The rate of inconclusive tests from the CDPH-PerkinElmer lab is seven and a half times higher than the other 22 COVID labs combined.
Of the labs with inconclusive results, state data reveals one out of every 256 results comes back inconclusive. One out of every 34 results from the CDPH-PerkinElmer lab is inconclusive according to data provided by CDPH.
ALSO READ: Are False Positives or Sensitive Tests Responsible For School COVID Spikes?
Whistleblowers Reach Out
After seeing our initial reports, a whistleblower reached out to CBS13 insisting that the continued high number of inconclusive results was not solely due to a problem with a chemical reaction or the highly sensitive test.
Since then, CBS13 has interviewed more than half a dozen whistleblowers, including several current and former employees at the lab.
They provided evidence of systemic problems inside the state lab and pointed to contamination, constantly changing protocols as well as unlicensed and inadequately trained staff as the reason for the high number of invalid and inconclusive results.
"That lab should be shut down because they're jeopardizing people's lives," said one whistleblower, whom we're calling Dottie.
Several came forward out of sheer concern for patient health, providing evidence of unlicensed lab techs learning basic skills on the job while processing patient samples.
The one thing all the whistleblowers had in common – they wanted the public to know what's actually happening inside your state lab.
QC Reports
CBS13 obtained dozens of quality control reports describing contamination, swapped samples, and repeated errors that could impact patients' test results.
Several reports indicate errors, including swapped specimen samples, that led to dozens of inaccurate results that were sent out to patients before the lab learned of the mistakes.
Several reports indicated that there was no system in place to immediately notify people that they received someone else's results. One report in mid-November stated, "A process for... retracting a report... needs to be established."
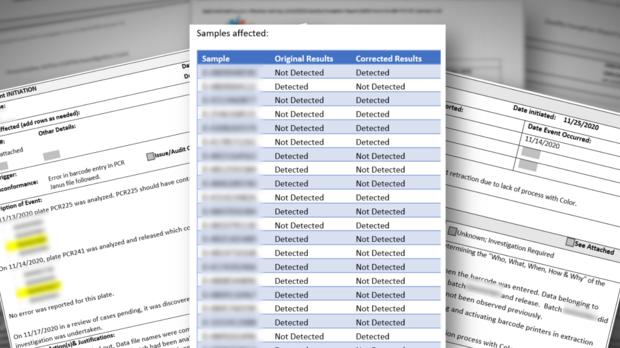
In response, CDPH acknowledged that at least "38 samples were reported incorrectly." The agency insisted that the patients were notified but could not provide evidence or an explanation of how those patients received corrected results.
UPDATE:
Two weeks after that initial response on Super Bowl Sunday, California Health and Human Services acknowledged in a press release that the lab had actually corrected 60 results.
Notably, the state's February 22nd press release stated that the state had found "significant deficiencies" at the lab back in December and PerkinElmer claimed they had "long since been resolved." It is not clear if CDPH's initial response was inaccurate or if the lab released another 22 wrong results during the two weeks following the initial CBS13 report.
The agency did not indicate how long it took the lab to notify patients that they had received the wrong results. The lab's Standard Operating Procedures for "Issuing Amended or Corrected Reports" was finalized and approved on February 8, 2021, the day after this initial story aired.
CDPH data indicates that the lab had processed more than 1.2 million samples in the three months before the lab had an official process in place to correct wrong results.
Sleeping & Restroom Swabs
To illustrate some of their basic concerns, several whistleblowers pointed us to an all-staff email that reported sample swabs were found in the restrooms more than once.
"Why would someone take a human specimen swab out of the lab into the bathroom?" CBS13 Investigative Reporter Julie Watts asked Dottie. "Who knows what they're doing? They're not supervised," she responded.
Whistleblowers also provided photos and videos of unlicensed lab techs watching videos on their phones and literally sleeping while processing the nasal swabs for testing.
However, they say the sleeping and swabs found in the restrooms are really just symptoms of systemic problems.
Systemic Problems
Whistleblowers point to a lack of qualified lab techs, a lack of qualified supervisors and constantly changing testing protocols.
For instance, they say, some of the techs who are extracting the crucial RNA from the sample, the part of the virus that is tested, are "unlicensed and fresh out of college."
While some techs don't have to be licensed, internal documents indicate that for months there was no licensed supervisor overseeing unlicensed techs on overnight shifts.
In one case, records indicate a licensed supervisor quit and was replaced with a "lead." According to staff records and publicly available information, the "lead" graduated with a Bachelor of Arts degree in 2019, is not a licensed Clinical Lab Scientist (CLS) and does not have the lab experience to act as a supervisor in a clinical lab.
"And the people that are training them are also unlicensed," Dottie pointed out. "When they don't have enough training, they don't know that what they're doing is wrong."
Several of the QC reports obtained by CBS13 describe errors that originated in the RNA extraction lab.
Emails reveal management asked supervisors to "re-train team members" months into the job, citing "several incidents where samples were lost."
According to state data provided in January, 205 samples were lost or spilled between November and January.
UPDATE: The state updated that number in its February 22nd press release and acknowledged to lawmakers that 252 samples could not be tested due to lab errors.
It is not clear if the state's initial data was incomplete or if an additional 47 samples were lost during the month of February.
Competency Documentation
Among the most concerning evidence provided by whistleblowers were emails from management indicating that lab techs had been processing patient samples before completing required training modules or getting signed-off for competency.
This was due, in part, whistleblowers say, to a lack of supervisors on some shifts. State and federal law require documented competency assessments that are performed by licensed supervisors.
In a written response, CDPH said, "All individuals who are working in the laboratory… handling specimens are credentialed and trained."
"This is FALSE!!!!!!!!!!!," one whistleblower said after seeing the CDPH response. "I was running samples my first day with zero clinical experience. So were the rest of my colleagues," the whistleblower insisted.
The agency acknowledged they did "identify a handful of individuals" who did not meet state requirements adding, "these individuals were moved to roles that align with their credentials or trained to ensure they now have the proper credentials."
Another whistleblower sent us a copy of the lab's "competency tracker" database. It indicated there were hundreds of lab employees that had still not been signed off for competency in one or more areas as of the end of January.
In response to the database, CDPH again insisted, "No employee without sign-off competency is running samples."
Whistleblowers dispute that claim.
UPDATE: Whistleblowers pointed us to an email that was sent to managers twenty-two minutes before CDPH provided that response on Super Bowl Sunday.
It indicates that managers were called that day to address the competency issues during the Super Bowl, "[s]ince this became even more urgent."
The email chain confirms that at least one manager was "headed in to help coordinate" citing a list of dozens of people in the RNA extraction lab alone who did not have competency as "a good place to start."
The competency tracker spreadsheet, which managers referred to in the email chain, contained the names of hundreds of employees who had not been signed off in one or more areas.
Experts Weigh In
CBS13 shared our findings with several industry experts including another state's laboratory director, federal regulators and state and federal laboratory industry groups. All were shocked by the evidence we shared.
Dora Goto, Governmental Affairs co-chair for the California Association for Medical Laboratory Technology (CAMLT) clarified that lab techs should not be processing human COVID samples if they don't have documented competency.
"It's federal law," Goto said.
Though Goto noted that there could be multiple reasons for the high number of inconclusive results coming out of this lab.
"That indicates to me that there are other systemic problems," Goto said.
As for sleeping on the job, Goto said, "these machines don't run by themselves. Regardless of how automated they are. You should be at least awake."
But she clarifies, while unlicensed, unsupervised lab techs sleeping on the job is certainly not a best practice, that would not necessarily be illegal in a public health lab.
For instance, she says, as long as there's a licensed supervisor in the building, state law doesn't technically require one physically overseeing unlicensed techs. However, she says it is standard practice.
Because the lab is operating as a public health lab, and because of the Governor's executive order, some licensing requirements are not as strict as they would be under normal circumstance for a clinical lab.
CONTINUING COVERAGE: CBS13 Investigates Problems at California's COVID Testing Lab
CDPH Response
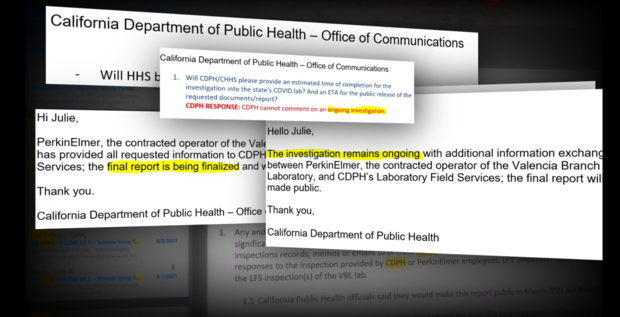
CDPH did not provide requested inspection records and did not comment on many of the specific allegations citing "personnel matters."
In an email response, the agency added:
"While we cannot discuss individual personnel matters, we take the safety and quality of the work in the laboratory seriously. PerkinElmer will take appropriate personnel action, including dismissing individuals who do not abide by these standards.
From the beginning, we made a commitment to iterate with our laboratory partner, PerkinElmer, to continuously improve our efforts to ensure that we are delivering the best results for the people of California. Human error is inevitable, but the laboratory strives for continuous improvement through training and process improvement."
Whistleblowers Want Reform
Dottie is one of a growing number of people quitting due to ethical concerns that, they say, were ignored by management.
"They're jeopardizing people's results," Dottie said. "That's why I left."
Whistleblowers said CDPH lab directors were among those who resigned from the lab, leaving only PerkinElmer representatives in top management positions.
In an email, CDPH said the lab initially had two, part-time, CDPH laboratory co-directors, but for "improved efficiency" they now have one full-time Perkin Elmer lab director.
Most of the whistleblowers we spoke with did not want to come forward publicly out of fear of retaliation.
They note that CDPH is not only the state agency responsible for inspecting and regulating labs, but it's also the agency that is responsible for issuing the Clinical Laboratory Scientist (CLS) licenses.
In short, the California Department of Public Health is the regulator and watchdog of its own state lab and employees.
Updated Response
After sending additional allegations to CDPH on Super Bowl Sunday, a representative from the California Health and Human Services Agency, which oversees CDPH, provided the following response:
"We are troubled by these allegations against our laboratory vendor and its employees and will investigate them immediately and thoroughly. If these claims are substantiated, they would represent a clear violation of our existing agreement with PerkinElmer and we would take swift action to fix these issues, up to and including termination of the contract.
"This lab was established by the state to provide accurate test results for the people of California as quickly as possible."
NOTE: This story was updated to include additional responses from the state and additional clarifying information from our continuing coverage.
WATCH THE CBS NEWS SACRAMENTO SPECIAL REPORT
THE COVID LAB: State Secrets Exposed
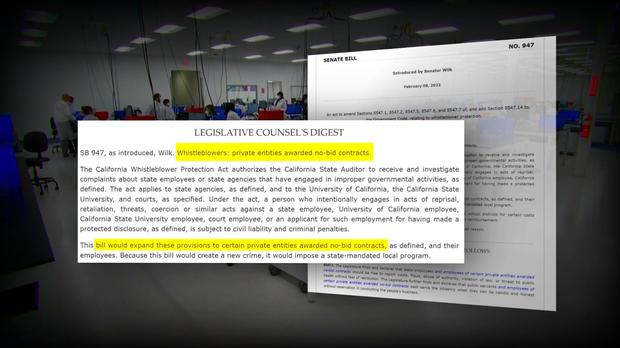
This 30-Minute Special Report is the culmination of 14 months of reporting that prompted state and federal investigations, resulted in two new pieces of legislation, and shined a spotlight on shocking public health failures that it appeared regulators tried to hide.
Following these reports, lawmakers introduced legislation that is intended to protect whistleblowers and ensure accountability and transparency long after the pandemic is over. The state ultimately terminated its $1.7B COVID contract with PerkinElmer.