Nature up close: Freezing soap bubbles
By "Sunday Morning" contributing videographer Judy Lehmberg.
Blowing soap bubbles has amused children (and adults) for centuries. Recently people have begun blowing soap bubbles in sub-freezing weather. Just this last November, the physics of water crystal formation in soap bubbles was explained at the American Physical Society - Division of Fluid Dynamics' 2017 meeting.
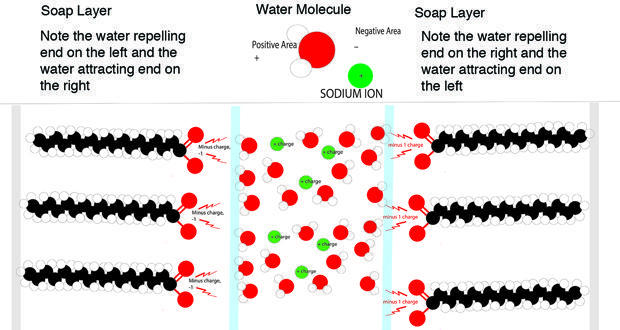
A little understanding of chemistry and physics is needed to understand freezing soap bubbles, and why water crystals move in the bubble. First, what is a soap bubble? It's formed from soap and water. As every schoolchild knows, water consists of hydrogen and oxygen, combined to form water. Water is a polar molecule, meaning that it has a slightly positive end (the hydrogen), which is attracted to the slightly negative (oxygen) end of another water molecule.
Soap is a long chain of carbon and hydrogen atoms linked together with hydrogen attached to the carbons. Soap molecules have a carbon-oxygen end that is negatively charged. A soap bubble consists of a layer of water, about one one-hundredth of a millimeter thick, sandwiched between two layers of soap molecules. The soap's negative end is attracted to the positive, hydrogen end of the water molecules.
Blow a soap bubble when the temperature is around -5 °F to 0 °F. The water between the soap layers begins to form crystals, which move around. As crystals form at the colder bottom of the soap bubble, heat is released as water changes from a liquid to its solid state, ice. The warmer water at the bottom of the bubble now has a lower surface tension, causing that water to move towards the top, carrying ice crystals with it. The crystals continue to dance and move until the whole bubble is frozen.
Watch time-lapse footage by videographer Becca Wood:
People living in cold climates can make these frozen beauties when the temperature hovers around 0°F. We've had the best success creating the bubbles by blowing a mixture of 1/6 cup clear dish washing detergent, 1 cup water, and 1/2 teaspoon of glycerin or corn syrup through a drinking straw. If you want to photograph the bubbles, in-direct light is best.
People living in warmer climates can try blowing soap bubbles onto a plate with ice on it, or in the freezer section of a refrigerator. Another really neat trick is to put some dry ice (frozen carbon dioxide) in a glass aquarium or cardboard box. Slowly blow a soap bubble above the dry ice. It will sink into the dense, cold carbon dioxide and freeze. Be careful handling the dry ice and don't touch it without gloves. Carbon dioxide freezes at -109 °F. Touching dry ice feels like touching a hot frying pan.
Judy Lehmberg is a former college biology teacher who now shoots nature videos.
For more info:
- Judy Lehmberg (Official site)
- Judy Lehmberg's YouTube Channel
To watch extended "Sunday Morning" Nature videos click here!