Melanoma drug breakthroughs stir "celebration" among skin cancer patients
(CBS/AP) Two new drugs have shown dramatic results against melanoma, researchers announced yesterday, giving people with the deadly skin cancer reason to cheer.
"This is really an unprecedented time of celebration for our patients," said Dr. Lynn Schuchter, of the University of Pennsylvania's Abramson Cancer Center. The new drugs are not cures, she said, but "the future is going to be to build upon the success" by testing combinations of these newer drugs.
In one study, an experimental drug called vemurafenib showed benefit so quickly against advanced melanoma that patients getting a comparison drug were allowed to switch after just months. The drug targets a gene mutation found in about half of all melanomas.
The second study tested Yervoy (ipilimumab), a drug that was just approved for newly diagnosed melanoma patients, and found it nearly doubled the number who survived at least three years.
The new studies were presented in Chicago on Sunday at the annual meeting of the American Society of Clinical Oncology and published online by the New England Journal of Medicine.
"This is a very exciting time in the field of melanoma," Dr. Perry Robins, president of the Skin Cancer Foundation, said in a written statement after the approval of Yervoy. "As melanoma incidence continues to rise, we are hopeful that this new therapy will extend life and improve the quality of life for patients with metastatic melanoma."
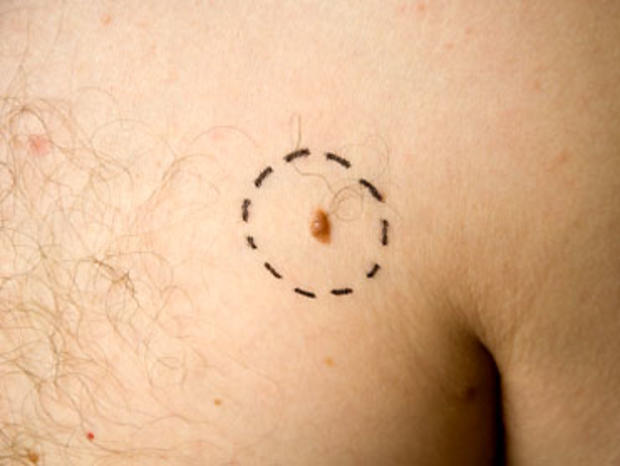
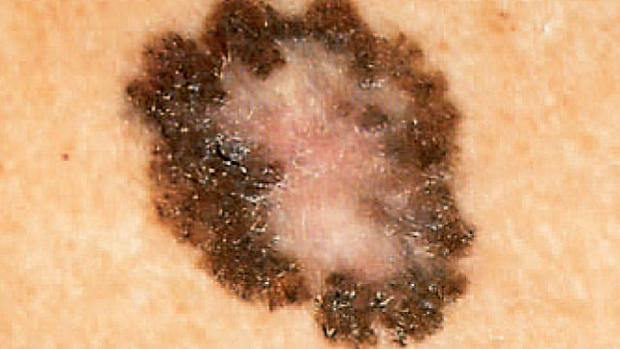
Melanoma is definitely on the rise. Last year there were 68,000 new cases and 8,700 deaths from it in the U.S., the American Cancer Society estimates. Only two drugs had been approved to treat it, with limited effectiveness, until Yervoy won approval in March.
Vemurafenib is aimed at a specific gene mutation, making it the first so-called targeted therapy for the disease. The drug got attention when a whopping 70 percent of those with the mutation responded to it in early safety testing. The new study, led by Dr. Paul Chapman of Memorial Sloan-Kettering Cancer Center in New York City, was the key test of the drug's safety and effectiveness. It involved 675 patients around the world with inoperable, advanced melanoma and the gene mutation. They received vemurafenib pills twice a day or infusions every three weeks of the chemotherapy drug dacarbazine.
After six months, 84 percent of people on vemurafenib were alive versus 64 percent of the others.
Less than 10 percent on the drug suffered serious side effects - mostly skin rashes, joint pain, fatigue, diarrhea and hair loss. About 20 percent to 30 percent of patients developed a less serious form of skin cancer.
The ongoing study is a landmark and the results are "very impressive" in people who historically have not fared very well, said Dr. April Salama, a Duke University melanoma specialist.
The study was sponsored by the drug's makers - Genentech, part of Roche, and Plexxikon, part of the Daiichi Sankyo Group - and many of the researchers consult or work for them. The companies are seeking approval to sell the drug and a companion test for the gene mutation in the U.S. and Europe. A Genentech spokeswoman said the price has not yet been determined.
The other new drug, Yervoy, is not a chemotherapy agent but a treatment to rev up the immune system to fight cancer. Dr. Jedd Wolchok of Memorial Sloan-Kettering led the first test of it in newly diagnosed melanoma patients. About 502 of them received dacarbazine and half also got Yervoy. After one year, 47 percent of those on Yervoy were alive versus 36 percent of the others. At three years, survival was 21 percent with Yervoy versus 12 percent for chemotherapy alone.
Side effects included diarrhea, rash and fatigue. More than half on the new drug had major side effects versus one quarter of those on chemotherapy alone.
Bristol-Myers Squibb paid for the study and many researchers consult or work for the company. Treatment with Yervoy includes four infusions over three months and costs $30,000 per infusion.
The Skin Cancer Foundation has more on melanoma.