FDA wants cancer warnings at indoor tanning salons
Cancer warnings are already on cigarette packs, and if a new proposal from the Food and Drug Administration goes through, they soon may appear on indoor tanning beds.
Indoor tanning salons could carry the new warnings and be subject to more stringent federal oversight, under the proposal unveiled by the FDA on Monday.
The FDA has regulated tanning beds and sun lamps for over 30 years, but for the first time ever the agency says those devices should not be used by people under age 18. The agency wants that warning on pamphlets, catalogs and websites that promote indoor tanning. And regulators are also proposing that manufacturers to meet certain safety and design features, including timers and limits on radiation emitted.
The FDA's order proposes to include a warning against use on people under 18 years old, and the labeling would have to include a warning that frequent users of sunlamp products should be regularly screened for skin cancer.
"Using indoor tanning beds can damage your skin and increase your risk of developing skin cancer," FDA Commissioner Dr. Margaret A. Hamburg, said in a news release. "The FDA's proposed changes will help address some of the risks associated with sunlamp products and provide consumers with clear and consistent information."
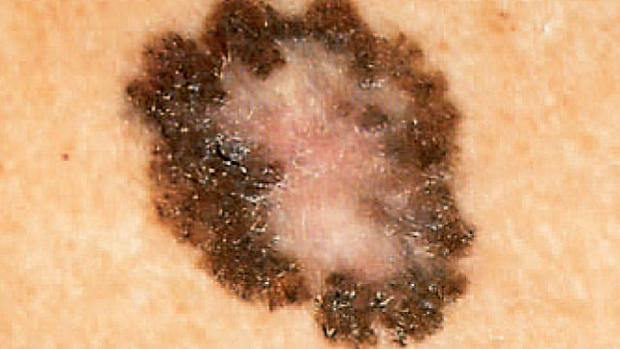
The government action is aimed at curbing cases of melanoma, the deadliest form of skin cancer, which have been on the rise for about 30 years. An estimated 2.3 million U.S. teenagers tan indoors each year out of about 28 million total people, but melanoma is the second most common form of cancer among young adults, according to the American Academy of Dermatology.
Recent studies have shown that the risk of melanoma is 75 percent higher in people who have been exposed to ultraviolet radiation from indoor tanning. While most cases are diagnosed in people in their 40s and 50s, the disease is linked to sun exposure at a young age.
Physician groups have been urging the U.S. government to take action on tanning beds for years, citing increases in the number of cases of skin cancer among people in their teens and 20s.
One group, the American Academy of Pediatrics, released a policy statement in March 2011 urging a ban on teens using tanning salons.
The American Academy of Dermatology's position statement says the medical group opposes indoor tanning and supports a ban on the production and sale of indoor tanning equipment for non-medical purposes.
"As a dermatologist I see the consequences of indoor tanning. I have to diagnose too many young people with melanoma and see the grief that it causes to these families," said Dr. Mary Maloney of the academy, on a call with FDA officials. Maloney said the FDA action is an important first step, but that her group would continue to push for a ban on the sale and use of tanning beds for people under age 18.
In February, a study of Missouri tanning salonsfound that 65 percent of 250 businesses surveyed would accept children ages 10 to 12, often without parental permission. The study was conducted by dermatologists at the Washington University School of Medicine in St. Louis, who at the time, called the research a "wake-up call" for parents in states that don't regulate tanning beds.
Currently the machines are classified as low-risk devices, in the same group as bandages and tongue depressors. The FDA's proposal would increase their classification to moderate-risk, or class II, devices. That would allow the FDA to review their safety and design before manufacturers begin selling them.
"They don't have to provide any data in advance before they go on the market, so we have no way of providing assurance that the tanning beds are performing up to specifications," said Dr. Jeffrey Shuren, FDA's director for medical devices.
Safety standards are important because recent studies show that many devices can cause sunburn even when used as directed. A 2009 study found that 58 percent of adolescents who tan indoors had sunburn exposure.
"If you get an indoor tan you shouldn't be burning," Shuren said.
- "Shocking" number of melanoma survivors tan, don't wear sunscreen: Study
- Study: Increased risk for young people and skin cancer
- Study: Tanning beds cause 170K skin cancer cases yearly
The agency said it will take comments on its proposal for 90 days before formulating a final regulation. Agency officials didn't give a timeframe for completion, but said it would be a priority.
The Indoor Tanning Association said it supported any changes that improve its customers' safety. But, in a statement, the group added that "we are concerned that these changes will burden our members with addition unnecessary governmental costs in an already difficult economic climate."