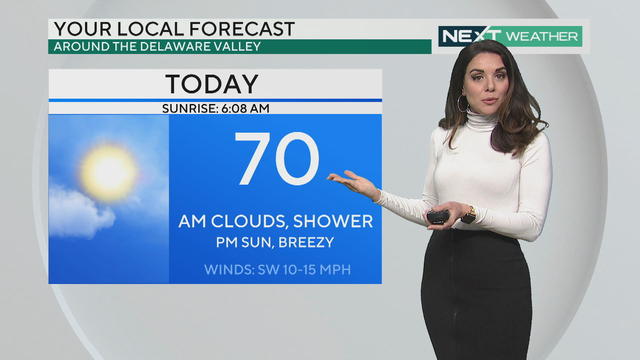
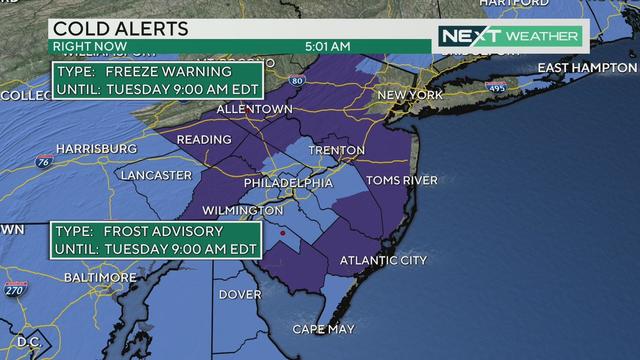
Philadelphia Health Officials Prepare For Child Vaccinations As Pfizer Seeks FDA Authorization For Children Under 12
Pfizer and BioNTech said Thursday they are seeking U.S. Food and Drug Administration emergency use authorization for their COVID-19 vaccine for children ages 5 to 11.